Selection of Yeasts for the Production of L-phenyl-acetil- carbinol Bybiotransformation in Shake Flasks

a–c Reactions catalyzed by pyruvate decarboxylase (PDC). a Pyruvate... | Download High-Quality Scientific Diagram

Improvement of the yeast based (R)-phenylacetylcarbinol production process via reduction of by-product formation - ScienceDirect

An N-methyltransferase from Ephedra sinica catalyzing the formation of ephedrine and pseudoephedrine enables microbial phenylalkylamine production - Journal of Biological Chemistry
Sciencemadness Discussion Board - Synthesis of Phenylacetylcarbinol by Alkyne Hydration and Subsequent Enamine formation - Powered by XMB 1.9.11

Synthesis of Chiral Catalyst Modifiers by Hydrosilylation of Cinchonidine and Their Application in the Hydrogenation of 1‐Phenylpropane‐1,2‐dione and Ethyl Pyruvate on a Supported Pt/Al2O3 Catalyst - Busygin - 2005 - European Journal

Asymmetric synthesis of ( S )-phenylacetylcarbinol – closing a gap in C–C bond formation - Green Chemistry (RSC Publishing) DOI:10.1039/C6GC01803C

Enhanced production of R-phenylacetylcarbinol (R-PAC) through enzymatic biotransformation - ScienceDirect
Raney-nickel-iron catalyst, its preparation and a method to produce L-norephedrine by hydrogenating L-phenylacetylcarbinol-oxime with said catalyst - Patent 2055379

Investigation of the l-phenylacetylcarbinol process to substituted benzaldehydes of interest - ScienceDirect

Synthesis of R-(−)-phenylacetylcarbinol by fermenting yeast and... | Download High-Resolution Scientific Diagram

DE60226038T2 - PROCESS FOR SYNTHESIS OF AMINES SUCH AS EPHEDRINE AND INTERMEDIATE PRODUCTS - Google Patents

Catalytic Cycle of Rhodium-Catalyzed Asymmetric 1,4-Addition of Organoboronic Acids. Arylrhodium, Oxa-π-allylrhodium, and Hydroxorhodium Intermediates | Journal of the American Chemical Society

EP1421055B1 - Methods for the synthesis of amines such as ephedrine and intermediates - Google Patents

Manufacturing by-products from, and stereochemical outcomes of the biotransformation of benzaldehyde used in the synthesis of methamphetamine - ScienceDirect

Biotransformation of benzaldehyde to L‐phenylacetylcarbinol (L‐PAC) by Torulaspora delbrueckii and conversion to ephedrine by microwave radiation | Semantic Scholar
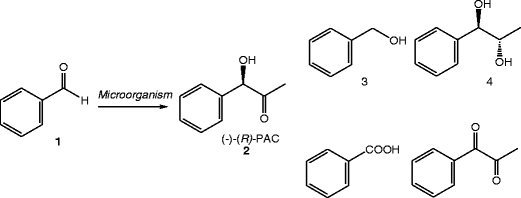
Potential of some yeast strains in the stereoselective synthesis of (R)-(−)- phenylacetylcarbinol and (S)-(+)-phenylacetylcarbinol and their reduced 1,2-dialcohol derivatives | SpringerLink

Potential of some yeast strains in the stereoselective synthesis of (R)-(−)- phenylacetylcarbinol and (S)-(+)-phenylacetylcarbinol and their reduced 1,2-dialcohol derivatives | SpringerLink
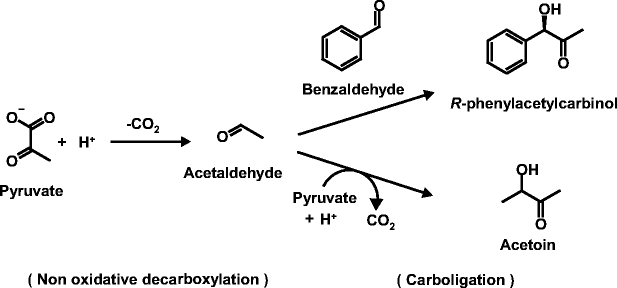
Enzymatic (R)-phenylacetylcarbinol production in a benzaldehyde emulsion system with Candida utilis cells | SpringerLink

US7176332B2 - Methods for the synthesis of amines such as ephedrine and intermediates - Google Patents