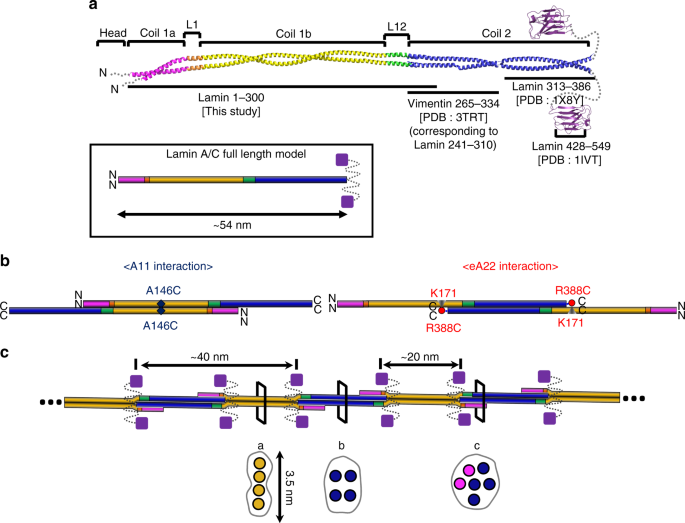
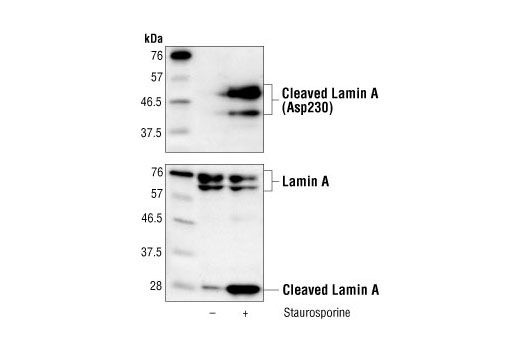
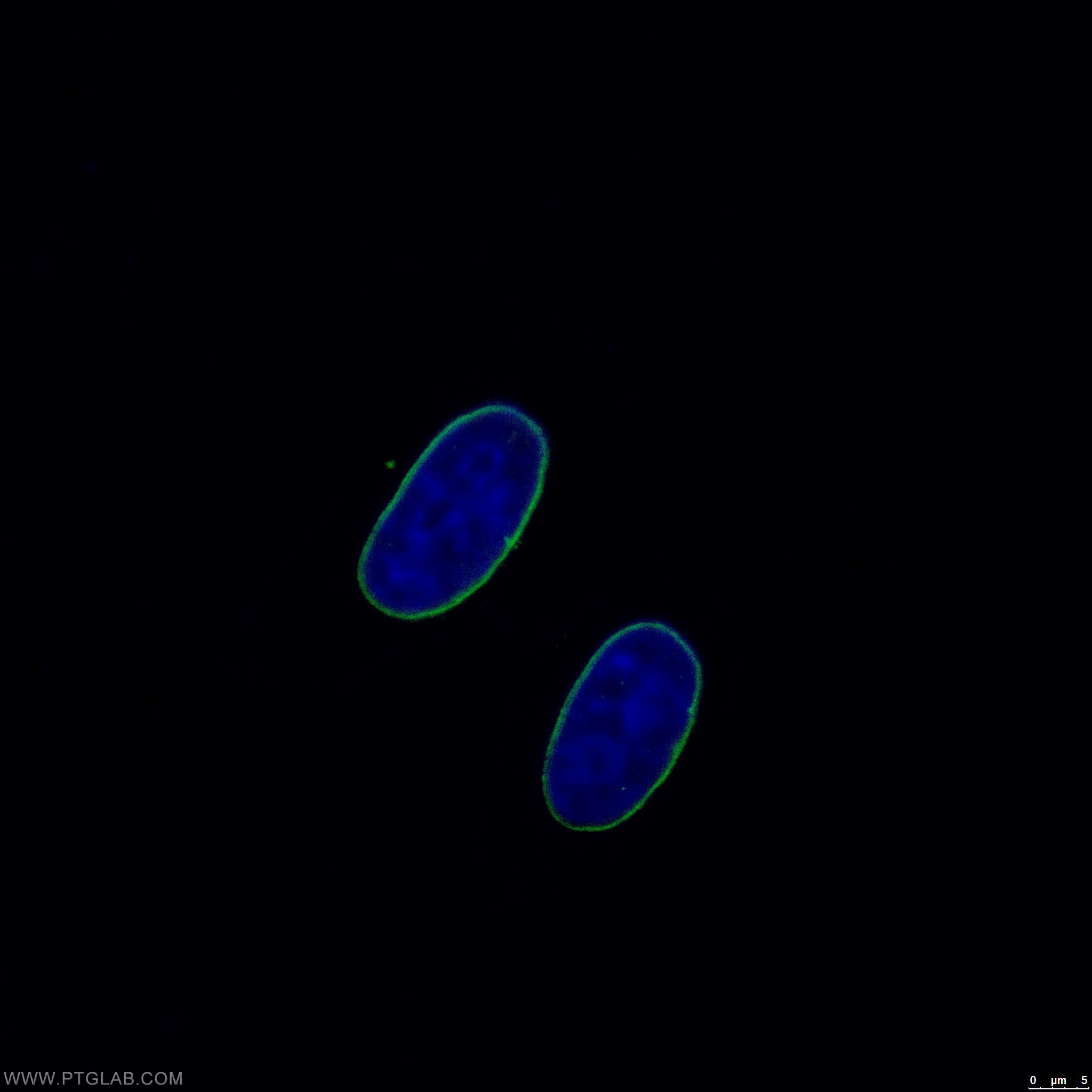
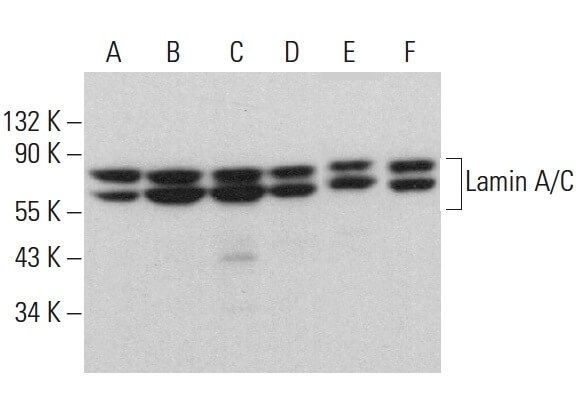
Missense Mutations in the Rod Domain of the Lamin A/C Gene as Causes of Dilated Cardiomyopathy and Conduction-System Disease | NEJM

Phosphorylated Lamin A/C in the Nuclear Interior Binds Active Enhancers Associated with Abnormal Transcription in Progeria - ScienceDirect
Lamin A/C Antibody, anti-human, REAdye_lease™ | Releasable fluorochromes | MACS Antibodies | Products | Miltenyi Biotec | Great Britain
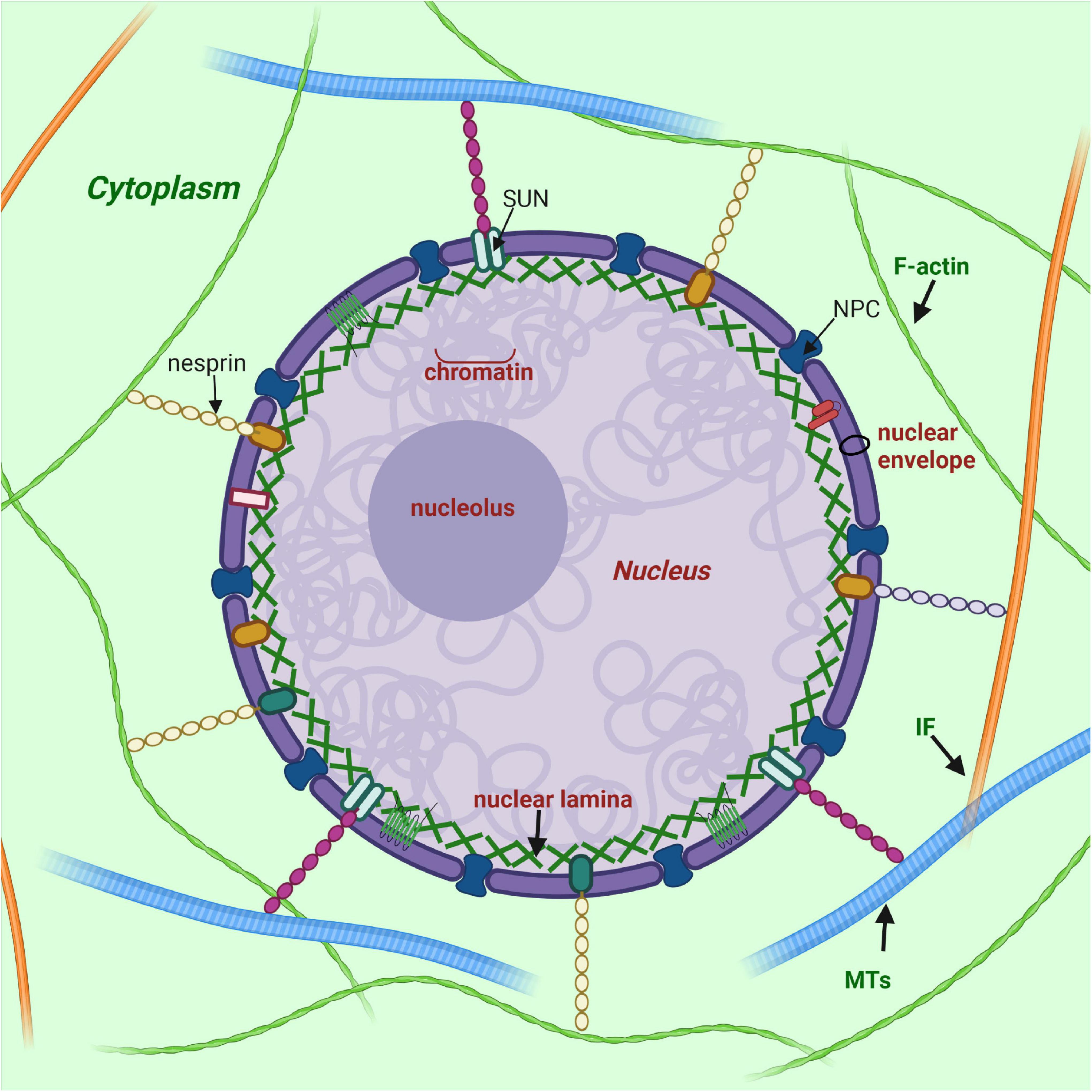
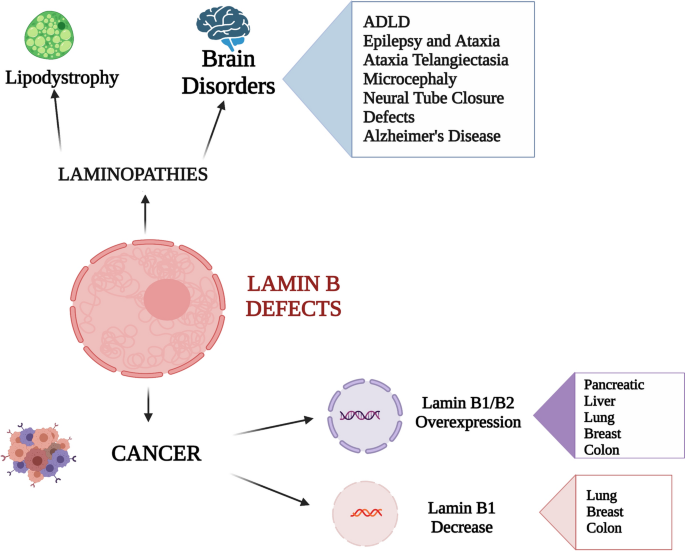
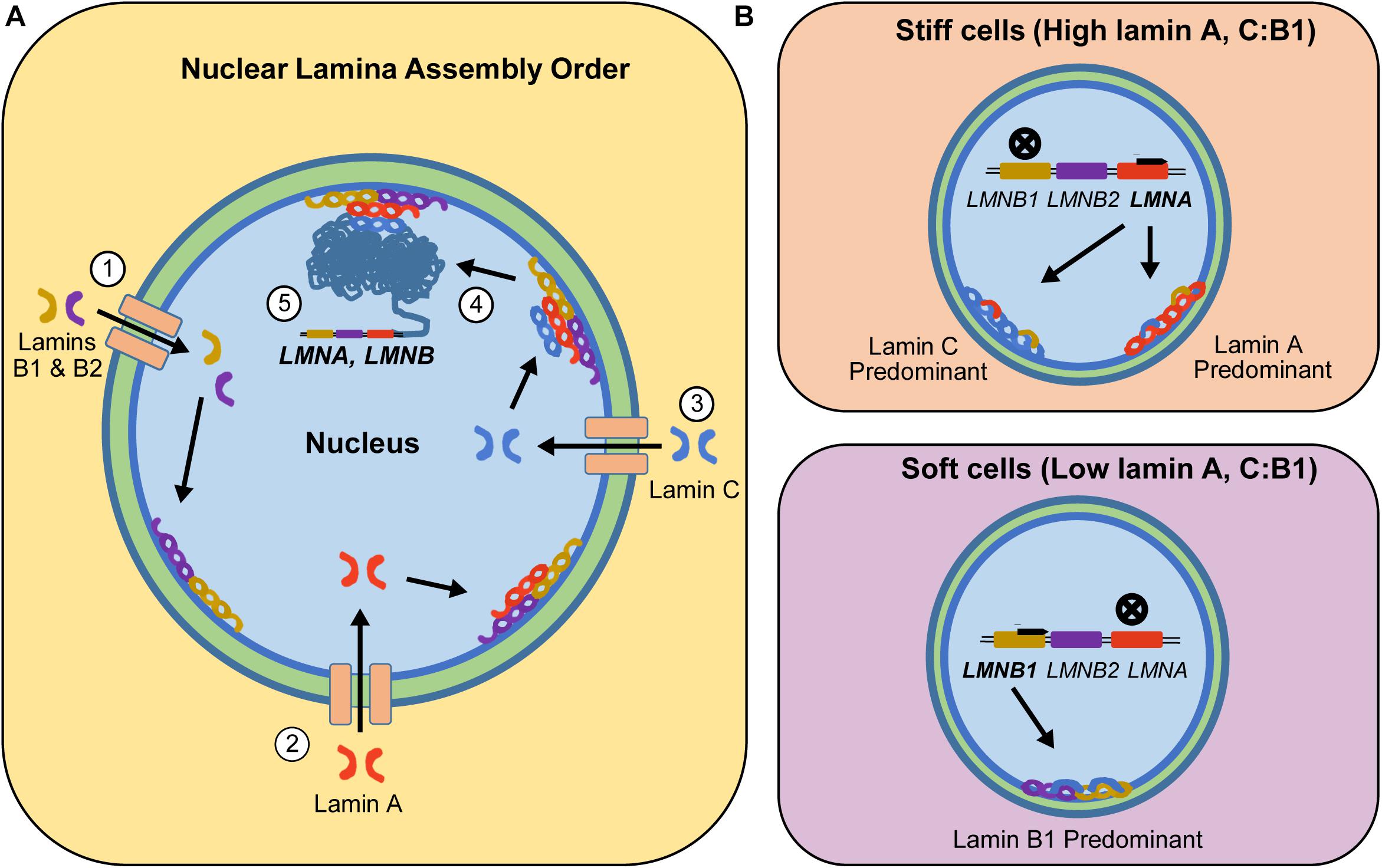
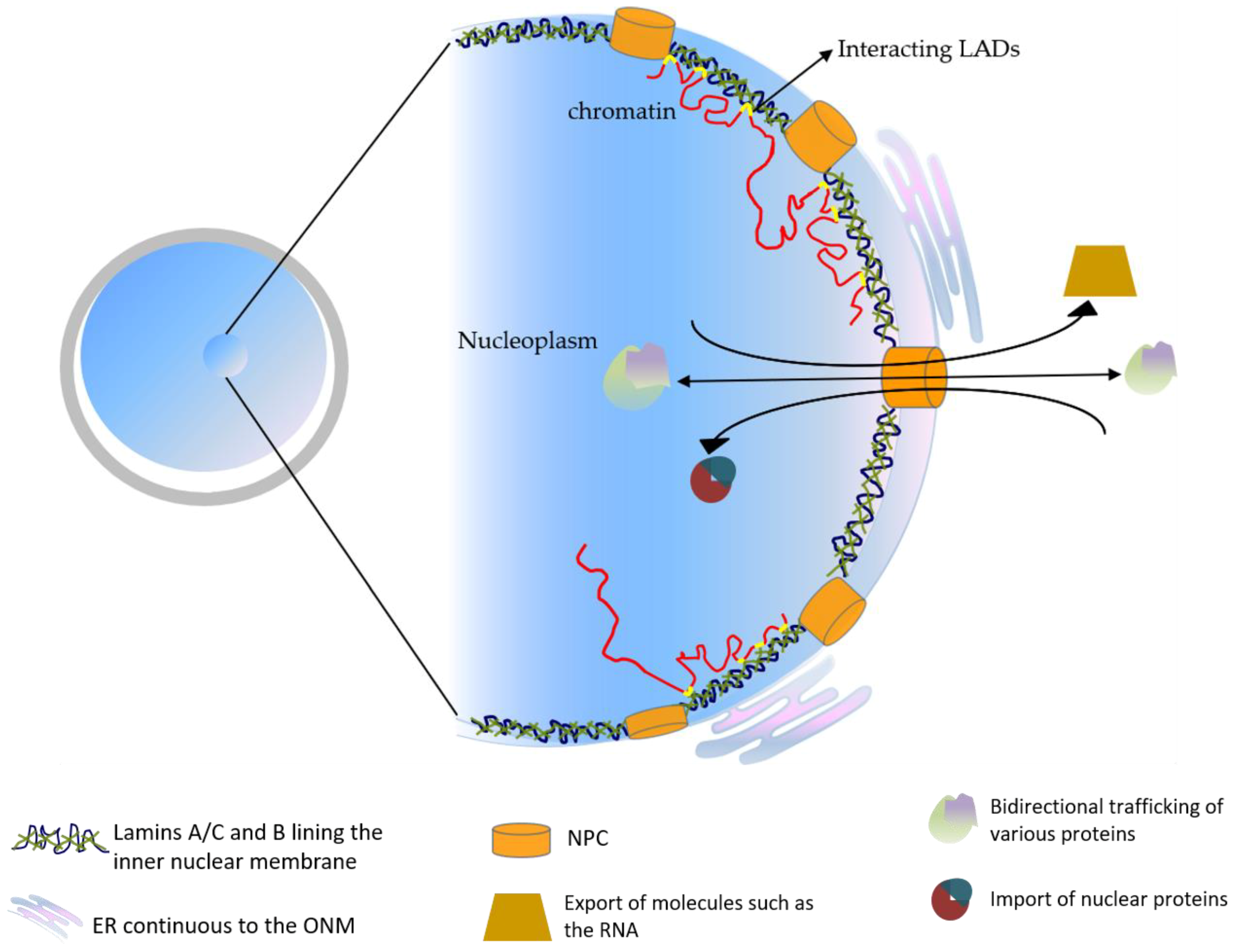
Frontiers | Diversity of Nuclear Lamin A/C Action as a Key to Tissue-Specific Regulation of Cellular Identity in Health and Disease

Nuclear envelope rupture and NET formation is driven by PKCα‐mediated lamin B disassembly | EMBO reports
Mutations in LMNA Modulate the Lamin A - Nesprin-2 Interaction and Cause LINC Complex Alterations | PLOS ONE

LAP2alpha maintains a mobile and low assembly state of A-type lamins in the nuclear interior | eLife

Lamin A/C Expression Is a Marker of Mouse and Human Embryonic Stem Cell Differentiation - Constantinescu - 2006 - STEM CELLS - Wiley Online Library
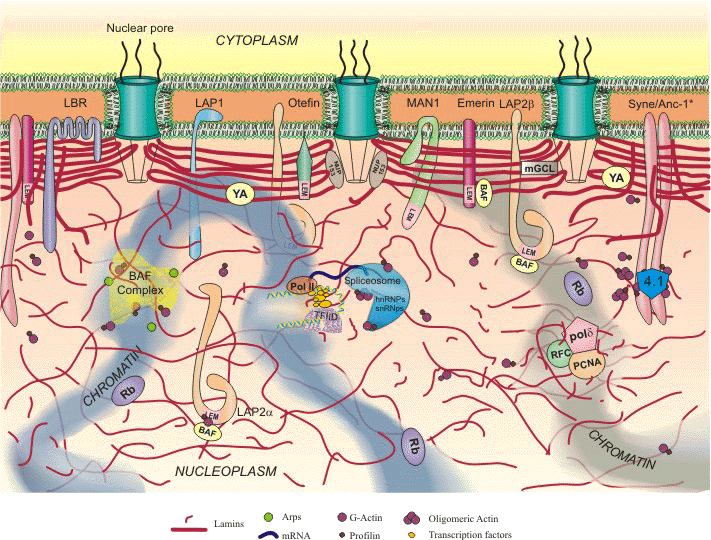
About Nucleoskeletal Intermediate Filament Proteins: The Nuclear Lamins: Goldman Lab: Feinberg School of Medicine: Northwestern University

Tyrosine phosphorylation of lamin A by Src promotes disassembly of nuclear lamina in interphase | Life Science Alliance