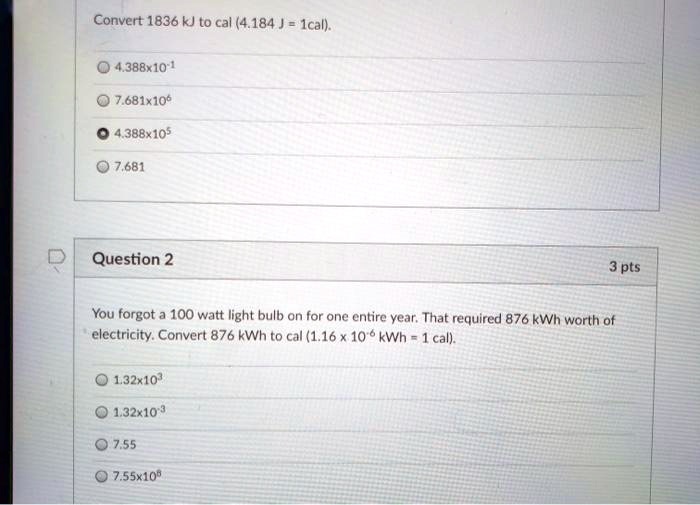
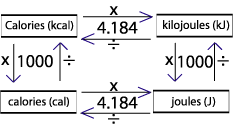
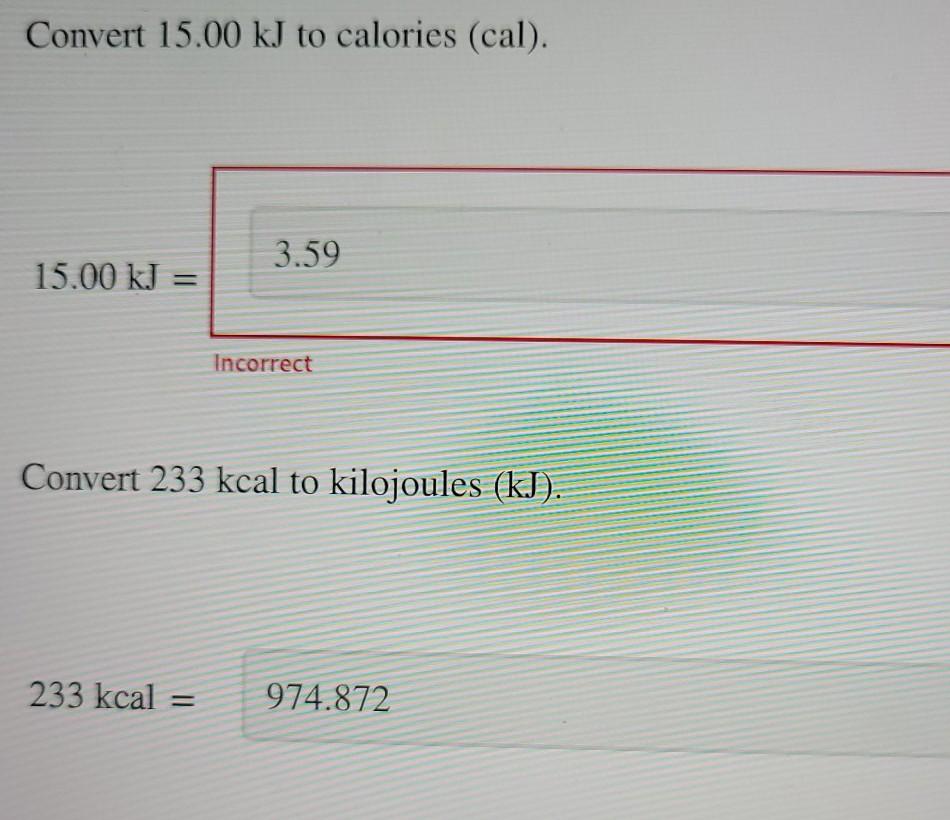
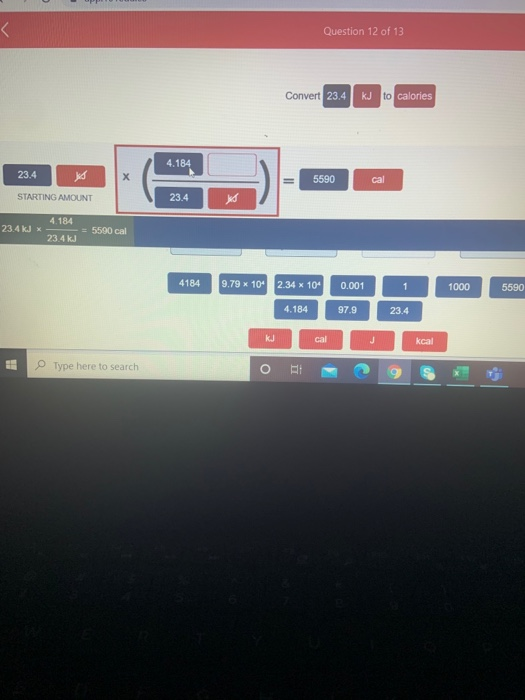
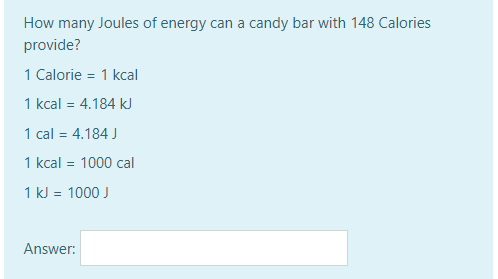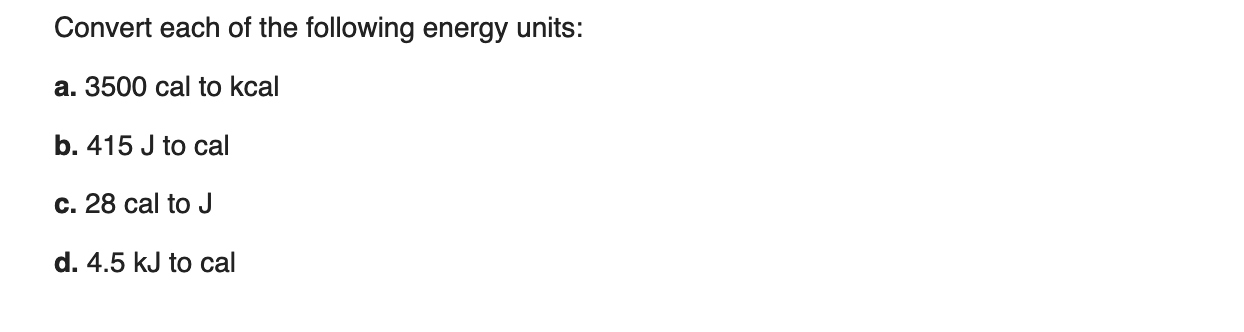Thermochemistry 1. The chemistry that measures the energy (HEAT) released or absorbed in a chemical reaction, or during melting or freezing, or condensing. - ppt download

Question Video: Conversion between Kilocalories and Kilojoules Using the Energy Consumption of the Adult Human Body | Nagwa